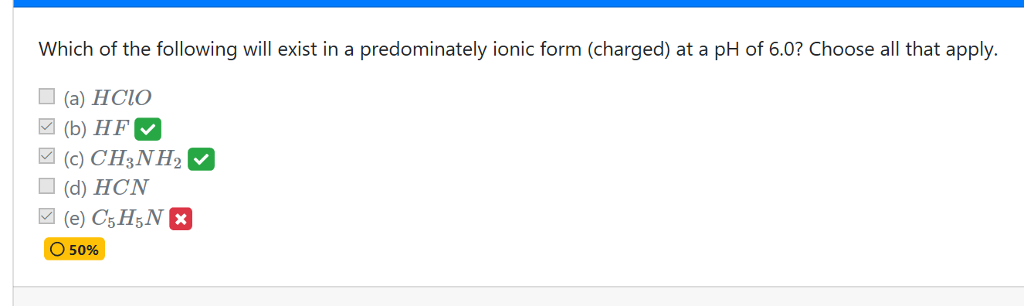
Which of the following will exist in a predominately ionic form (charged) at a pH of 6.0? Choose all that apply. (a) HClO (b) HF (c) CH3NH2 (d) HCN O 50% Show transcribed image text
Expert Answer
An answer will be send to you shortly. . . . .

Which of the following will exist in a predominately ionic form (charged) at a pH of 6.0? Choose all that apply. (a) HClO (b) HF (c) CH3NH2 (d) HCN O 50% Show transcribed image text
An answer will be send to you shortly. . . . .