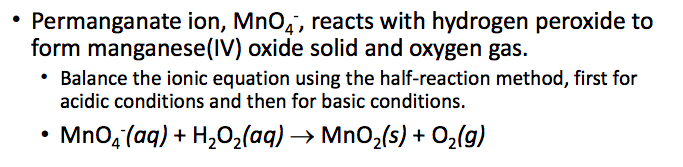 MnO4 – (aq) + H2O2 (aq) ? MnO2(s) + O2 (g)
MnO4 – (aq) + H2O2 (aq) ? MnO2(s) + O2 (g)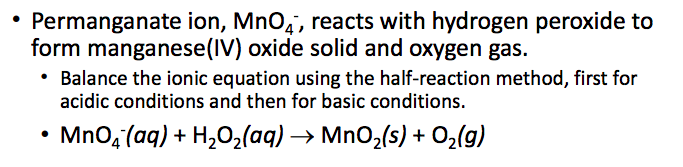
Permanganate ion, Mno, reacts with hydrogen peroxide to form manganese(IV) oxide solid and oxygen gas. Balance the ionic equation using the half-reaction method, first for acidic conditions and then for basic conditions . MnQ(aq) + H2O2(aq) ? MnO2(s) + O2(g) Show transcribed image text
Expert
OR
PayPal Gateway not configured
OR
PayPal Gateway not configured
PayPal Gateway not configured
PayPal Gateway not configured